[Disclaimer: I am not a doctor, and this is not medical advice. I’m writing this to process my vaccine timing and choice decision, as well as feelings about the world slowly opening up.]
If you’ve been reading this blog, you will know that I got my first vaccine shot a couple of months ago, and that it was AstraZeneca. At the time, I mentioned that there were some known issues with blood clots, and the incidence was estimated to be about 4 per million by the United Kingdom Medicines and Healthcare Products Regulatory Agency. With more data, this number has been changed. The Canadian National Advisory Committee on Immunization (NACI) has now estimated that the VTT blood clot rate could go to as high as 1 in 55,000, with increased observation time. So far (as of June 4th), there have been 2,346,032 doses of AstraZeneca administered, and there have been 50 cases reported to PHAC[1] or Health Canada, including 31 with laboratory results showing VITT, including 6 deaths. This 50/2.3 million is about one in 47,000, 31/2.3 million is about one in 76,000, so one in 55,000 seems like a reasonable estimate. The death rate is about one in 390,000, similar to the ~19/9.5 million (or one in 500,000) we discussed last time. (Please also note that the NACI is now recommending that people watch for VITT for up to 52 days post-AstraZeneca dose.)
That being said, with more evidence from Canada and elsewhere, the NACI is now saying that:
“Due to the observed AstraZeneca safety profile and risk of VITT, offering an alternative product with a more acceptable safety profile and expected comparable immunogenicity profile, while enabling individuals to make an informed choice is ethically justifiable. This is expected to lead to increased accessibility and acceptability for those who were initially offered a first dose of the AstraZeneca vaccine, including those who are most at risk of COVID-19.”
In this, they are weighing the apparent increased risk of death from VITT against the dangers to an un-immunized individual (and of an un-immunized population), and the expected timeline for mRNA vaccine availability vs. the increased risks of COVID variants that are resistant to a single dose of a vaccine.
As always, your decision should be made in consultation between you and your doctor. I’m planning to get an mRNA vaccine (Pfizer or Moderna) for my second dose, but I don’t know when, yet. Likely within the next month or two.
So, with the NACI saying that:
– If you got Pfizer or Moderna for your first shot, you should get the same (or the other) for your second shot (strong recommendation)
– If you got AstraZeneca for your first shot, you can get AstraZeneca, or Pfizer, or Moderna for your second shot (and that they understand why people might prefer Pfizer or Moderna to AstraZeneca for their second shot)
The next question is ‘when do you get your second shot?’
There are a few factors at play here:
– The total local number of COVID cases
– The number of cases of more dangerous variants (and wanting to stamp the total local number of cases so as to avoid evolving more dangerous variants)
– Vaccine type & availability
– The waiting time for maximum effectiveness (and whether a third shot will be necessary/allowed/possible/etc…)
– The total local number of COVID cases:
Currently, in Ontario, the number of cases per day is trending downwards by 20-40%, week on week:
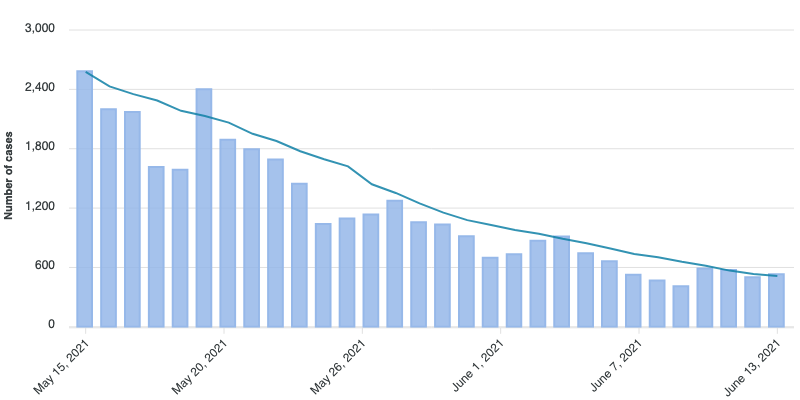
This is great news! However, there is also disturbing news that a new variant, ‘Delta’ (B.1.617.2), with about 60% greater transmissibility is starting to take over. (Some more Ontario-specific news on the Delta variant.) Specifically, it is estimated that while one vaccine dose is 60-80% effective against ‘wild-type’ COVID, and two doses are >85%, against the Delta variant, one dose is only about 30% effective, and it requires two doses to be about 80% effective.
– The number of cases of more dangerous variants (and wanting to stamp the total local number of cases so as to avoid evolving more dangerous variants)
So, people might want to get their second dose as soon as possible, in order to protect as much as they can against the new Delta variant (and any others that may arise). (This has the added side-benefit of reducing the total number of cases, and reducing the number of chances that COVID-19 has to mutate into other variants.)
– Vaccine type & availability
From the NACI: “Canada is anticipating large supplies of mRNA vaccines in the summer months that will be sufficient to complete the second dose in all age groups for whom immunization is recommended.”
So, availability of mRNA vaccines seems to no longer be a concern.
– The waiting time for maximum effectiveness (and whether a third shot will be necessary/allowed/possible/etc…)
So, here’s where it gets tricky. Because all of these vaccines are super-new, there is a limited amount of data on exactly when the ‘sweet spot’ is as far as how far doses should be spaced from each other for maximum effectiveness. Also, the presence of variants and vaccine mixing complicates matters.
The current recommendation from NACI is:
– Pfizer: 21 days to 16 weeks
– Moderna: 28 days to 16 weeks
– AstraZeneca: 28 days to 16 weeks
(The minimum number of days are based on general scientific understanding of how vaccines work, along with the number of days between doses in the clinical trial. The maximum number of days was based on ’emerging evidence of the protection provided by the first dose of a two-dose series’ and ‘limited COVID-19 vaccine supply and ongoing pandemic disease’.)
Based on increased vaccine availability, Ontario is now starting (Monday June 14th) to book second shots for those who had AstraZeneca for their first shot 8 weeks after their first shot (down from 12 weeks).
Ontario is also echoing the NACI recommendations:
“If your first dose was:
– AstraZeneca: you can get AstraZeneca, Moderna, or Pfizer for your second dose when you are eligible and it’s at least 12 weeks after your first dose.
– Moderna or Pfizer: you should get the same vaccine for your second dose when you are eligible and it’s at least 28 days after your first dose. You can switch between Moderna and Pfizer safely if the original vaccine you got is not readily available.”
“With informed consent, individuals can choose between a second dose of AstraZeneca or an mRNA vaccine, at an eight to 12-week interval, recognizing that while waiting 12 weeks helps to ultimately provide more protection, some may choose to receive their second dose sooner to have the increased protection provided by a second dose earlier. All of these options provide protection against COVID-19, including the Delta variant, and have been deemed safe.”
So, with some digging, I found this paper: “Single Dose Administration, And The Influence Of The Timing Of The Booster Dose On Immunogenicity and Efficacy Of ChAdOx1 nCoV-19 (AZD1222) Vaccine“, which talks about how the amount of time between vaccine doses correlates with overall effectiveness. This study includes data from the UK, Brazil, and South Africa (about 17 thousand participants, about half receiving the vaccine, half control). The upshot is that the effectiveness of the second dose goes from about 55% at <6 weeks to >80% after >12 weeks, with indications that the enhanced effectiveness starts to kick in somewhere between 9 and 11 weeks.
So, knowing all of this, which vaccine should you get for your second shot, and when? As I said above, this decision should be made between you and your doctor, but given the data above, it would make sense to weigh your perceived day-to-day danger level (along with mounting dangers from Delta and other variants) against the knowledge that waiting a bit longer could give you greater immunity overall. I’ll be 8 weeks out from my first immunization this coming weekend, and I can see myself waiting a couple of weeks after that for my second shot (which I intend to be a mRNA vaccine). (There is also some evidence that waiting 12 weeks leads to better mRNA vaccine effectiveness in older people.)
I’m still making my decision, though, as it may be that the extra overall vaccine effectiveness may not be meaningful, as the overall effectiveness may be ‘high enough’ (especially in reducing or eliminating serious COVID effects), and the extra security from emerging variants (and getting to enjoy more of the summer) may be worth it.
Let me know what you think for your own situation.
As always, thanks for reading, and stay safe!
P.S. If you want a more in-depth discussion of how the immune system works, and why vaccine mixing makes sense, let me know in the comments below! I had thought that this would be necessary to explain NACI’s vaccine mixing recommendation, but I think their ethical arguments are more directly relevant at the moment. (And this post is already quite long.)
P.P.S. Some more references I glanced at but only used for background:
These have a whole bunch of interesting graphs that I didn’t have time to get into:
https://www.publichealthontario.ca/-/media/documents/ncov/epi/covid-19-epi-confirmed-cases-post-vaccination.pdf?la=en
https://www.health.gov.on.ca/en/pro/programs/publichealth/coronavirus/docs/vaccine/COVID-19_AstraZeneca_Vaccine_admin.pdf
Here are a bunch of articles that I didn’t have time to get into:
https://www.cbc.ca/news/health/canada-pandemic-turning-point-covid-19-1.6027497
https://www.canada.ca/en/public-health/services/publications/healthy-living/canadian-immunization-guide-part-1-key-immunization-information/page-10-timing-vaccine-administration.html
https://globalnews.ca/news/7920859/ontario-astrazeneca-second-doses-moderna-pfizer/
https://news.ontario.ca/en/release/1000329/ontario-accelerates-second-doses-of-astrazeneca-covid-19-vaccine
https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)32623-4/fulltext
https://www.nature.com/articles/d41586-021-00785-7
https://www.nature.com/articles/d41586-020-03326-w
https://www.nature.com/articles/d41586-021-00001-6
https://www.ctvnews.ca/health/coronavirus/how-effective-is-the-astrazeneca-covid-19-vaccine-what-you-need-to-know-1.5349237